Water is an essential ingredient not just for life but also in home, cosmetics, and personal care products. Its quality can make all the difference in the quality of your products. How are you monitoring your water? If you are not testing your water systems regularly, you could miss lurking harmful microorganisms like Burkholderia cepacia. It isa waterborne microorganism that can take up residence in your water purification systems and become a reoccurring problem in your facility. Set yourself up for the best chance for success for high quality water if you commit to routine testing of your different water sources. This process will aid your facility and allow quality control professionals to assess and analyze any changes. Data gathering is critical to helping you to reach a state of control of your water system and reduce potential risk of missing harmful microorganisms that could get into your products. Although routine water system monitoring is not required in the personal care industry, water remains one of the top contamination sources found in manufacturing, so it is important that you are testing your system using validated methods on a regular basis.
What is Burkholderia cepacia?
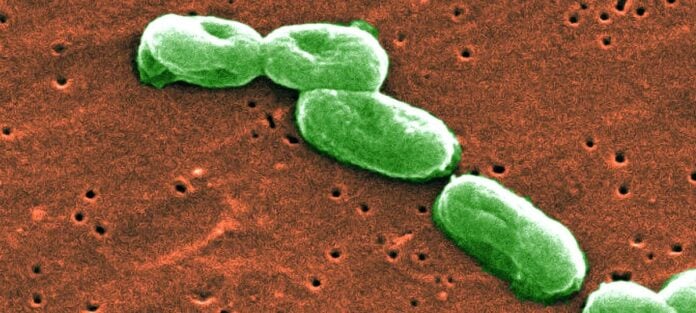
Burkholderia cepacia is a microorganism species that is part of a larger group called the Burkholderia cepacia complex (BCC). There are 23 species in this group, and they are all Gram-negative, rod-shaped bacteria that have been shown in recent years to be of concern for home and personal care product manufacturers. Some species belonging to the BCC are opportunistic pathogens. The BCC has been directly linked to product recalls and in some cases consumer illness.
With this risk to consumers in mind, regulatory bodies tasked with protecting public health, such as the US FDA, have taken notice and issued numerous recalls for products that were found to be contaminated with BCC microorganisms. Non-sterile water-based products have featured prominently in BCC-related recalls, including products such as sanitizers, nasal sprays, mouthwashes, skin creams, electrolyte solutions, adult and baby wipes, and laundry detergent. Following an FDA advisory in 2017, USP introduced chapter 60 for the detection of BCC in non-sterile aqueous products in 2019. Currently, there is no official guidance in the European Pharmacopeia or International Organization for Standardization (ISO) on specific testing for BCC.
BCC is especially troublesome because of its ability to resist preservatives and antimicrobial agents, growing even in unfavorable environments. One species in particular, Burkholderia multivorans, has been noted to grow well in environments with little food such as distilled water. A case study shared by the FDA examined a situation in which Burkholderia multivorans was detected in a product prior to market release. Two batches of preserved nasal spray were found to be contaminated during routine microbial testing. They dug deeper in their investigation and started testing older lots of product and surprisingly found some lots also contaminated with BCC, even though they previously had tested negative. How could this be happening? To make it even more difficult, BCC species are generally slow growing microorganisms. The company found that the bacteria were initially inhibited by the preservative but then overcame it and flourished, making their initial negative tests inaccurate. Detailed testing identified the microorganism, not just as a member of theBCC, but as Burkholderia multivorans, which allowed the company to link contaminated batches to each other and find the source of the contamination. Ultimately, the organism was traced back to a Burkholderia multivorans biofilm that had formed in the purified water system and having established the cause, they were able to correct the plumbing and continue with additional corrective actions.
Another reason why it is important to identify the BBC species is that one preventive measure is not necessarily sufficient to eradicate all member species, as they are unique and may respond differently to remediation attempts. Some species like Burkholderia multivorans can cause additional concern and require different action or a broadened scope of investigation. With that in mind, manufacturers have begun to revisit their microbiological testing protocols to detect BCC contamination. Not all members of the complex are designated as pathogens so it’s important to accurately identify to the species-level. However, this has proven problematic using traditional DNA sequencing methods and has required advances in microbial identification strategies.

Going beyond the species
There are times when you need to go beyond the species level. In an investigation, you may find multiple contamination events that identified the same species. Did they come from the same origin? Strain typing is a microbial characterization technique that can help distinguish members of the same species to the strain level using well-established, highly accurate DNA sequence analysis. For example, a company identifies a microorganism of the same species in two separate samples, one was from environmental monitoring of the water system and the other was recovered from routine quality testing of a finished product. If the microbial strains are identical, according to strain typing, that means that their DNA is the same and the microorganism contaminating the finished product is the same one you’re finding in your water. This is the reason why strain typing is critical to resolving and remediating contamination events. Strain typing has been traditionally done by analyzing multiple coding genes in independent steps using Multi-Locus Strain Typing (MLST). Now, there is a technology that is being utilized called Next Generation Sequencing (NGS) that can analyze the entire DNA genome in one test producing the same results in less steps.
Test early and regularly for microbial contamination
Catching microbes, like BCC, before they become a problem and working to implement a microbial contamination control strategy means monitoring your environment with accurate technology. Testing earlier and regularly in the manufacturing process and implementing good manufacturing practices can reduce contamination in your raw materials, water, and finished products. These practices ensure that products are consistently produced and controlled according to quality standards designed to aid in the quality control process.
Any time part of a facility is offline due to a microbial contamination event, it equates to lost profits. If the contamination originated from your water system, no amount of cleaning the manufacturing vessels will solve the problem because that is not the source. An accurate species level identification and, when necessary, strain typing can help put you on the right path to finding the microorganism source.

How Can Charles River Laboratories Help You?
The relative cost of accurate microbial IDs is small especially compared to the loss of inventory or, worst case, the cost of a recall. Charles River Labs recognizes that accurate and reliable microbial identification data is critical not only for certain organisms such as the Burkholderia cepacia complex, but for all microbial isolates that are part of quality control practices. Our unique combination of Celsis® rapid microbial detection and Accugenix® microbial identification keeps your manufacturing operations running efficiently and smoothly. Explore our portfolio of integrated services at www.criver.com/cosmetics.